If the liquid molecules are strongly attracted to the tube molecules, the liquid creeps up the inside of the tube until the weight of the liquid and the adhesive forces are in balance. For many fluids, the tangential or shearing stress that causes the flow is directly proportional to the rate of shear strain. 3.2 FACTORS AFFECTING VISCOSITY The viscosity of Newtonian fluids is affected by temperature, pressure, and, in the case of solutions and mixtures, by composition. The reciprocal of viscosity is known as fluidity and is the measure of the ease of flow. In simple words, it is the opposition to the flow. The video below shows several different long chained oils, each progressively more viscous.\). What is Viscosity Viscosity is defined as the internal resistance of the fluid. It is also the resistance to the flow of a liquid. Glycerol, CH 2OHCHOHCH 2OH, is viscous partly because of the length of the chain but also because of the extensive possibilities for hydrogen bonding between the molecules. A viscous fluid, such as heavy crude oil which is too viscous to enable it to be pumped from a flowing phase of a reservoir into and along a pipeline for. Coefficient of viscosity may be defined as the force of friction required to maintain a velocity difference of 1 cm/sec between two parallel layers, 1 cm. Viscosity is defined as the force of friction between the successive layers of a flowing liquid. Fuel oil, lubricating grease, and other long-chain alkane molecules are quite viscous for this reason. This is because the molecular chains get tangled up in each other like spaghetti-in order for the liquid to flow, the molecules must first unravel. 
To recognise viscosity as a useful property of liquids. Liquids containing long molecules are invariably very viscous. Learning objectives To understand that viscosity is a measure of a liquids resistance to flow. The higher the VI, the more stable the viscosity. The lower the VI, the more the viscosity is affected by changes in temperature. Being able to measure the viscosity will allow to predict the behavior in a process or an. It is mostly used to characterize the viscosity-temperature behavior of lubricating oils. It can be defined as a liquids internal resistance to flow. Honey, mostly glucose and fructose (see image below) is a good example of a liquid which owes its viscosity to hydrogen bonding. The viscosity index (VI) is an arbitrary, unit-less measure of a fluids change in viscosity relative to temperature change. Liquids whose molecules are polar or can form hydrogen bonds are usually more viscous than similar nonpolar substances.  
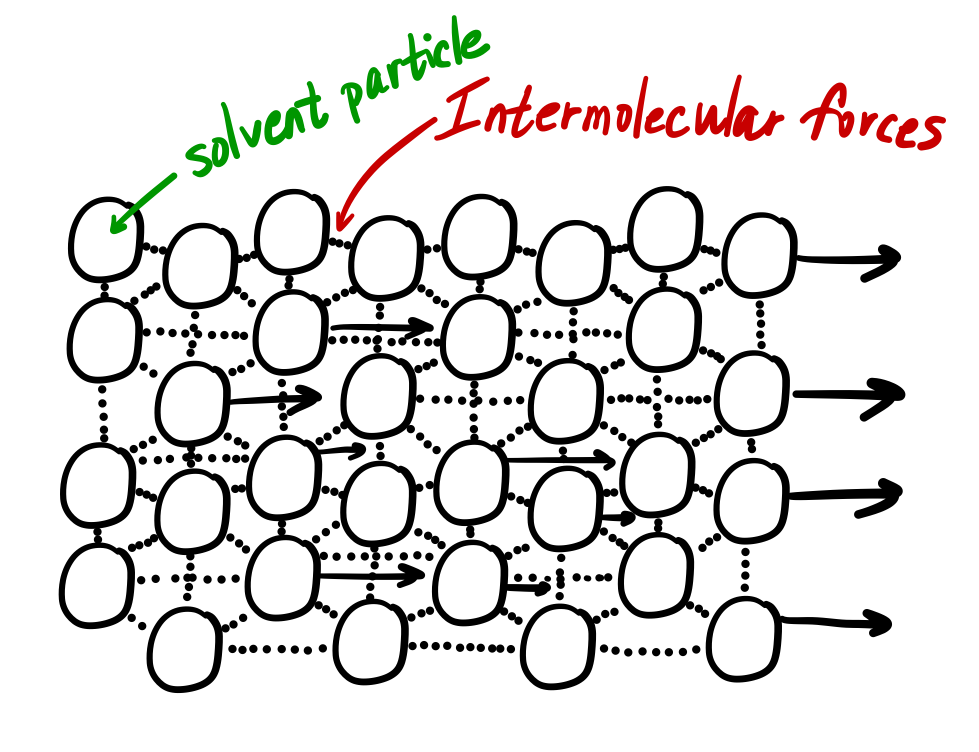
The viscosity of water at 20 ☌ is 1.0020 millipascal seconds (which is conveniently close to one by coincidence alone). Viscosity is governed by the strength of intermolecular forces and especially by the shapes of the molecules of a liquid. The force necessary to move a plane of area A past another in a fluid is given by Equation 2.2.1 2.2.1 where V V is the velocity of the liquid, Y is the separation between planes, and is the dynamic viscosity. Viscosity is first and foremost a function of material. Coefficient of Viscosity: The force of friction required to maintain velocity difference of 1cm. This arises due to internal friction between layers of fluids as they slip past one another while liquid flows. Those like ether or gasoline which flow very readily have low viscosities. Viscosity: Viscosity is a measure of resistance to flow of liquids. Liquids which flow very slowly, like glycerin or honey, have high viscosities. The resistance to such flow is called the viscosity. \)īecause its molecules can slide around each other, a liquid has the ability to flow.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
